Working group Singh
Transcriptional Reprogramming in Cancer Plasticity Lab
Why is pancreatic cancer research so important?
The 10-year survival rate for patients with pancreatic cancer (PDAC) stands at approximately 1%. While mortality rates for other cancers have significantly declined in recent years, PDAC is projected to become the second leading cause of cancer-related deaths by 2030. In Germany, between 20,000 and 22,000 individuals are diagnosed with PDAC each year, with almost an equal number succumbing to this complex disease during the same timeframe. The incidence and mortality rates of PDAC are closely linked to aging, a major concern in many European nations where populations are rapidly aging. The hallmarks of PDAC include early tumor invasion, with over 80% of cases displaying local or distant metastasis at the time of diagnosis. Unfortunately, among the 20% of patients who undergo surgical resection, many experience local or distant tumor recurrence due to the presence of resilient micro-invasive tumor cells. Currently, the primary treatment option for all PDAC patients consists of surgical resection followed by intensive chemotherapy. However, one-third of these patients experience tumor relapse within three years. Given the limited scope of personalized therapeutic approaches, there is an urgent need for advanced molecular screening-based individualized stratification to enhance overall prognosis in PDAC patients.
Key biomedical challenges in the prognosis and treatment of pancreatic cancer
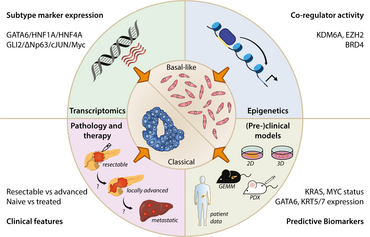
The intricate molecular heterogeneity present in tumor and stromal immune compartments poses challenges for conventional therapies, contributing to a grim prognosis and complicating patient stratification for treatment. The identification of transcriptomic subtypes has significantly improved early prognosis and therapeutic interventions for PDAC patients. For instance, transcriptomic profiling has revealed two distinct subtypes: 'classical' and 'basal-like.' The basal-like subtype is strongly associated with poorer prognosis and resistance to therapy, while the classical subtype correlates with a more favorable outlook. For further information, please refer to our latest review article published in Trends in Cancer 2022 (PMID: 36117109).
How can we understand the disease complexity of pancreatic cancer?
Our Max-Eder research group focuses on advancing personalized medicine and translational cancer research. We believe a deeper understanding of tumor and immune cell differences could improve patient outcomes in PDAC. Our latest study shows how different tumor subtypes interact with immune cells in the tumor environment to drive PDAC aggression. Specifically, we found that the characteristics of tumor cells attract inflammatory macrophages, which push away immune cells that could attack the tumor and make the disease worse. For more details, please refer to our publication in Nature Cancer 2021 (PMID: 35122059), its preview in Cancer Discovery 2022 (PMID: 34862194), and in Nature Communications 2025 (PMID: 39762215).
Additionally, metastatic pancreatic cancer (PDAC) has a poor outlook, with a 5-year survival rate below 3%. We have recently discovered a new factor, the ROBO3 receptor, that helps the cancer spread and become resistant to treatment. By blocking ROBO3 signaling, we can reduce fluid buildup and liver metastases, and restore sensitivity to chemotherapy. This study uncovers an important mechanism behind the aggressive subtype of PDAC and supports treatment strategies that aim to target these subtypes to overcome resistance. For further details, please see our publication in JCI Insight 2022 (PMID: 35993361).
Research focus
- Exploration of cross-talk between inflammatory immune components and tumor cells in pancreatic cancer progression and chemoresistance
- Identification of lineage-specific transcription factors and chromatin modifiers as potential biomarker for the patient stratification in pancreatic cancer
- Establishment of epigenetic-based therapies for precision treatment in pancreatic cancer
Press Releases

Our publications featured on the journal cover page
Funding
Awards and Honors
2026
- Young Oncologist Award (1st Prize) in the Postdoctoral Scientist category awarded to Dr. Lukas Klein, Bratislava, Slovakia
- Young Oncologist Award (2nd Prize) in the PhD Student category awarded to Rebecca Samuel, Bratislava, Slovakia
- Young Oncologist Award (3rd Prize) in the Postdoctoral Scientist category awarded to Dr. Xueyen Wu, Bratislava, Slovakia
2025
- Xueyen Wu was awarded magna cum laude for her PhD thesis, UMG
- Mildred-Scheel Endowed Professorship awarded to Shiv K. Singh by Deutsche Krebshilfe Xueyen Wu was awarded magna cum laude for her PhD thesis, UMG
- Dr. Lukas Klein received the prestigious Mildred-Scheel Postdoctoral Fellowship from Deutsche Krebshilfe
2024
- Best Talk Award to Xueyen Wu at the Molecular Medicine UMG Retreat, Einbeck, Germany
- Laura Urbach was awarded magna cum laude for her PhD thesis, UMG
- Best Poster Award to Laura Urbach at the German Pancreas Club (DPC), Bonn, Germany
2023
- Lukas Klein was awarded summa cum laude for his PhD thesis, UMG
- Laura Urbach received a DAAD scholarship travel award to attend a special AACR Conference on Pancreatic Cancer, Boston, USA
- Lukas Klein received a DAAD scholarship travel award to attend a special AACR Conference on Pancreatic Cancer, Boston, USA
2022
- Successful extension of the Max-Eder Research Grant by Deutsche Krebshilfe
- Niklas Krebs was awarded summa cum laude for his MD thesis, UMG
- Göttingen Very Important Publication Award (Gö-VIP) for a JCI Insight paper (PMID: 35993361)
- Niklas Krebs received the DFG Clinician Scientist Fellowship for research abroad at Princess Margaret Cancer Centre, Toronto, Canada
- Best Medical Thesis Award to Niklas Krebs, UMG
2021
- Mengyu Tu was awarded magna cum laude for her PhD thesis, UMG
- Poster Award to Lukas Klein at the CERFA Cancer Symposium (virtual), Spain
2020
- Hans-Chiari Excellence Award for the best scientific talk awarded to Lukas Klein at the German Pancreas Club (DPC), Halle, Germany
- Best Poster Award to Niklas Krebs at the German Pancreas Club (DPC), Halle, Germany
- Travel Award to Niklas Krebs and Lukas Klein at the German Pancreas Club (DPC), Halle, Germany
2019
- Mildred-Scheel Fellowship awarded to Shiv K. Singh at the 9th Mildred-Scheel Cancer Conference, Bonn, Germany
- Best Poster Award to Niklas Krebs at the European Association for Cancer Research (EACR), Berlin, Germany
- DGVS MD Research Fellowship awarded to Niklas Krebs by the Deutsche Gesellschaft für Gastroenterologie, Verdauungs- und Stoffwechselkrankheiten (DGVS)
2018
- Best Poster Prize awarded to Niklas Krebs at the German Pancreas Club (DPC), Ulm, Germany
- Max-Eder Research Grant awarded to Shiv K. Singh by Deutsche Krebshilfe, enabling the establishment of an independent research group at UMG (2018)
Team
Principal Investigator
Prof. Dr. Shiv K. Singh
e-mail address: shiv.singh(at)med.uni-goettingen.de
Team Members:
Dr. rer. nat. Lukas Klein (Postdoc)
Xueyen Wu (PhD student, CSC fellow)
Rebecca Samuel (PhD student)
Paul Stenke (MD Student)
Jian Shi (MD Student)
Frederike Penz (Research Assistant, BTA)
Nehir Sahin (PhD Student)
Alumni
Dr. Laura Urbach (PhD thesis)
Dr. Niklas Krebs (MD thesis)
Dr. Mengyu Tu (PhD thesis, CSC fellow)
Daniela Grimm (MSc thesis)
Eva Schmitt (MSc thesis)
Lena Wieland (MSc thesis)
Marcel Romey (MSc thesis)
Çezarela Hoxha (MSc thesis)
Ekta Sidhu (MSc thesis)
William Tan (BSc thesis)
Clarissa Lohmann (BSc thesis)
Leona Schüürhuis (BSc thesis)
Henrike Hasecke (BSc thesis)







